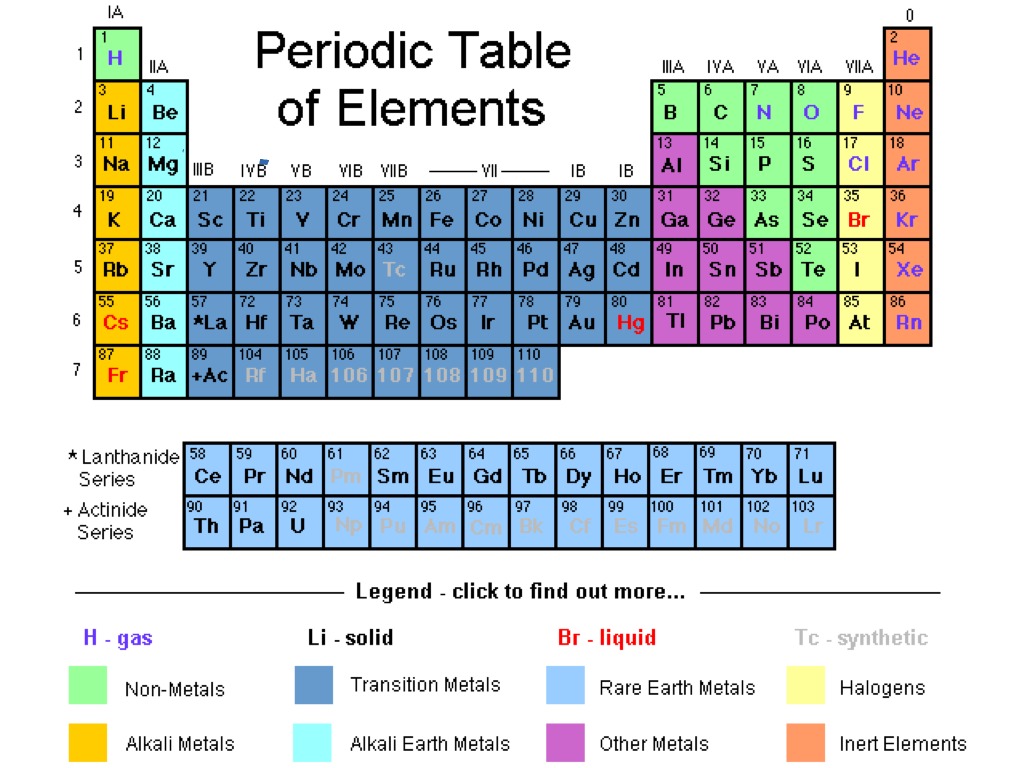
 Group 2 elements are chemical elements having their outermost element pair in s optionalīeryllium ,magnesium, calcium, strontium,barium, and radiumįorm cation by removing one valence electrons 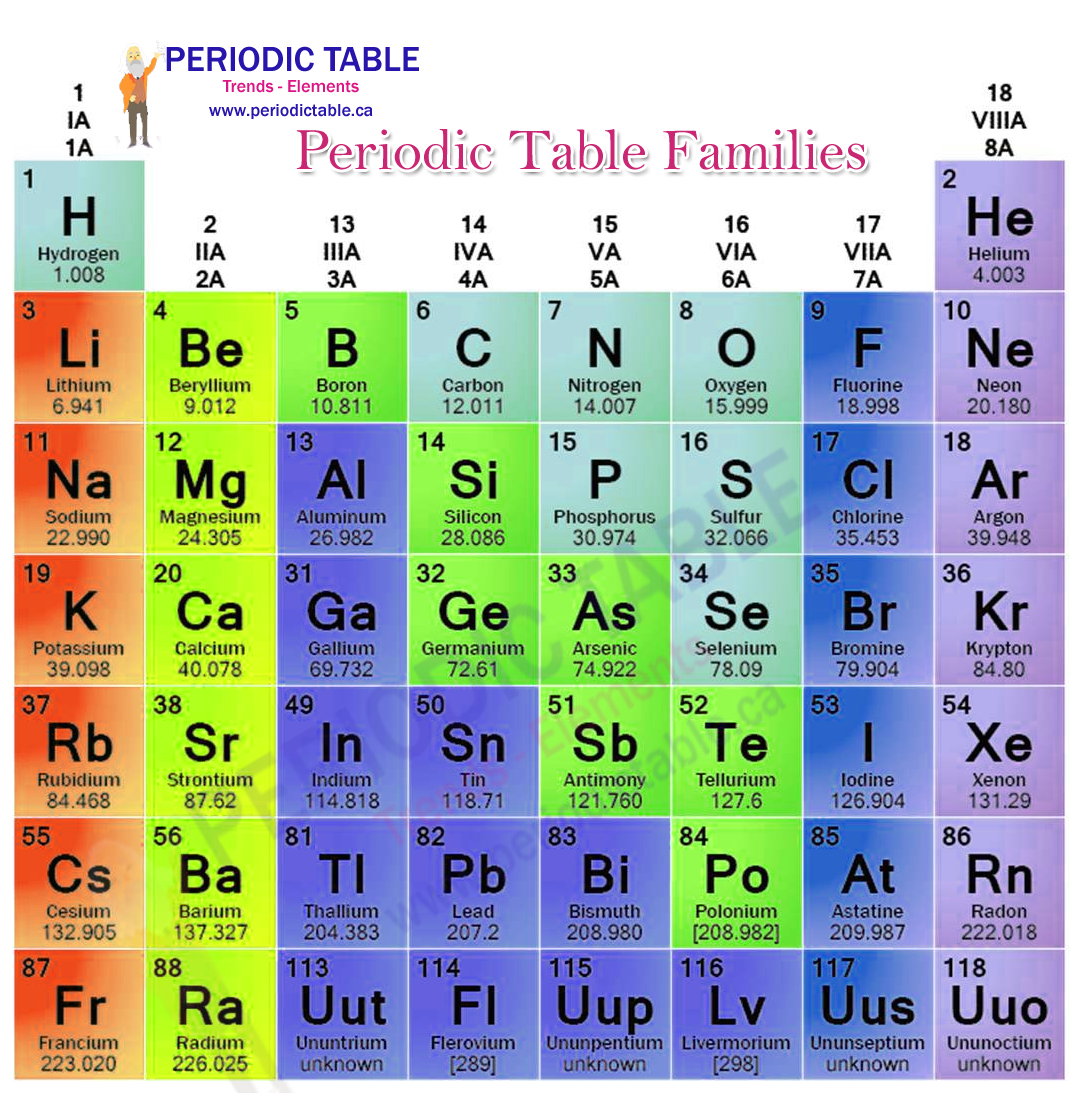
Group 1 elements are chemical elements having an unpaired electron in outermost s orbital The below infographic shows a lot of comparisons relating to the distinction between group 1 and group 2 elements. The key distinction between group 1 and group 2 elements is that all group one elements have unmatched electrons in their outermost orbital, whereas group 2 elements have paired electrons in their outer orbital. The group 1 and a pair of dissents from one another depending on the amount of electrons in their outer orbital. Radium (Ra) What’s the difference? Elements of the periodic table in groups 1 and 2 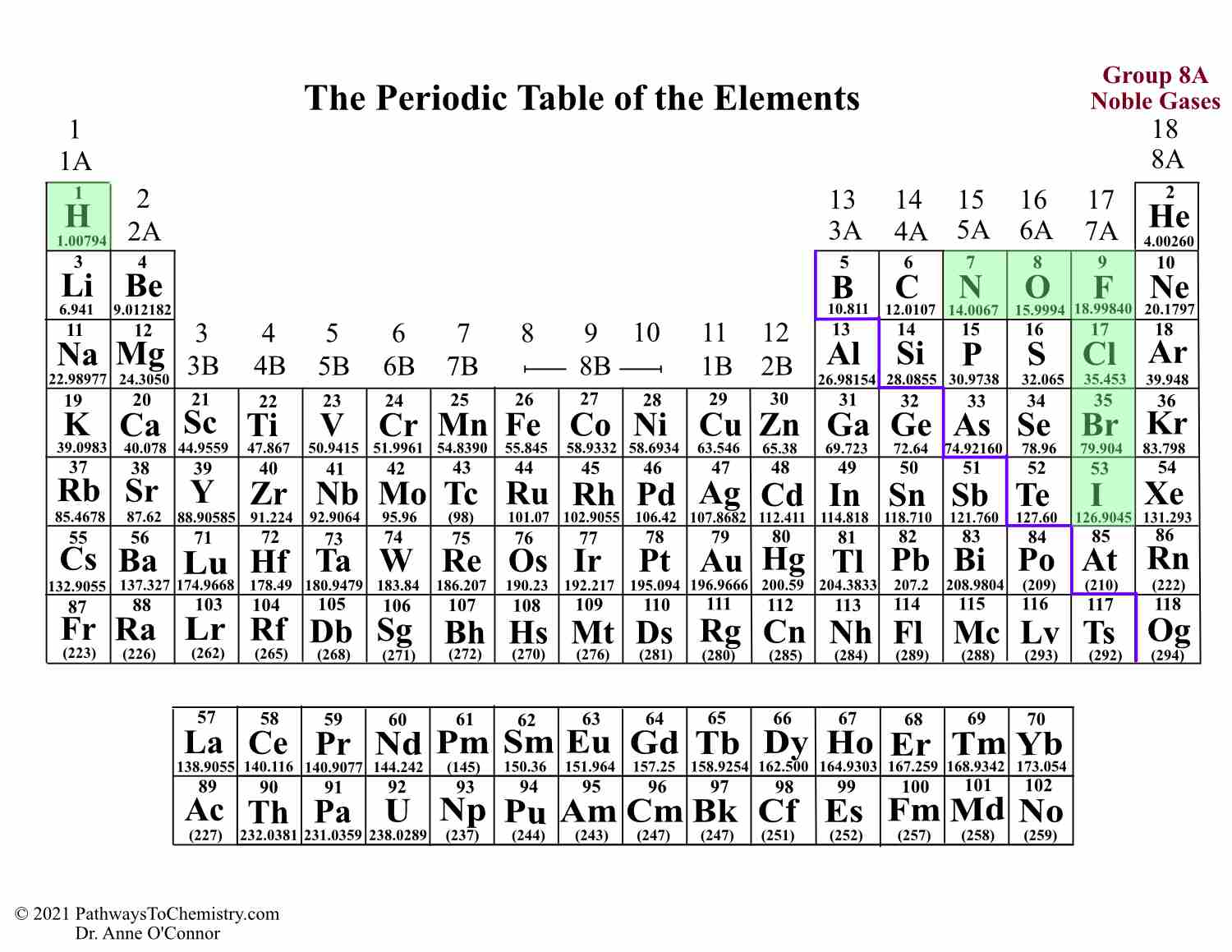
The members of this cluster area unit as follows: We name them as alkaline-earth metal metals. Further, this group is the second column of the s block. Therefore, their valence electrons are in the form of ns2. Group 2 elements are chemical elements having their outermost electron pair in an s orbital. What are the characteristics of Group 2 Elements of Periodic table
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
